Laser-tissue Interaction
Introduction:
The variety of interaction mechanisms that may occur when applying laser light to biological tissue is manifold. Specific tissue characteristics as well as laser parameters contribute to this diversity. Most important among optical tissue properties are the coefficients of reflection, absorption, and scattering which have been discussed in detail in previous modules. Together they determine the distribution of light and total transmission of the tissue at a certain wavelength. This initial distribution of light lies at the very heart of all ensuing effects whether they are therapeutic, or diagnostic. Thermal tissue properties such as heat conduction and heat capacity will be added in this module. On the other hand, the following parameters are given by the laser radiation itself: wavelength, exposure time, applied energy, focal spot size, energy density, and power density. Among these, the exposure time is a crucial parameter when selecting the type of interaction as will find shortly.

Schematic of laser-tissue interactions
During the first decades after invention of the laser by Maiman (1960), many studies have been conducted investigating potential interaction effects by using all types of laser systems and tissue targets. Although the number of possible combinations for the experimental parameters is unlimited, mainly three different types of interaction mechanisms are classified today: photochemical, photothermal and photomechanical.
Each of these interaction mechanisms will be discussed in detail in this module. In particular the physical principles governing these interactions are reviewed.
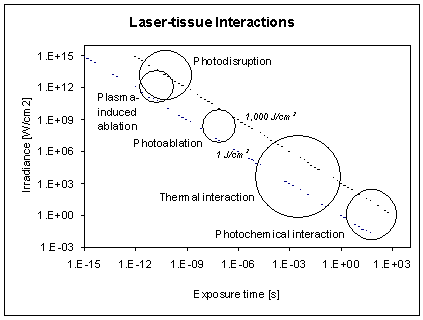
Before going into detail, an interesting observation deserves to be stated. All these seemingly different interaction types share a common property: the characteristic radiant exposure [J/cm2] ranges from approximately 1 J/cm2 to 1000 J/cm2. This is surprising since the irradiance itself varies over more than 15 orders of magnitude. Thus a single parameter distinguishes and primarily controls these processes: the duration of the laser exposure which is largely similar to the interaction time itself.
A log-log plot of irradiance versus pulse duration with the basic interaction mechanisms is shown in the figure below. In this figure the photomechanical mechanisms are separated in three sub categories: photo-ablation, plasma-induced ablation, and photodisruption. In the graphs the two diagonals show constant radiant exposures at 1 J/cm2 and 1000 J/cm2, respectively. According to this graph, the time scale can roughly be divided in three major sections: continuous wave or exposure times > 1 s for photochemical interactions, 100 s down to 1 ms for photothermal interactions, and 1 ms and shorter for photomechanical interactions. It should be clear, however, that these boundaries are not strict and adjacent interaction types cannot always be separated. Thus overlap in these main regions do exist. For example, in the range of 1 ms to several hundreds of ms, the interaction mechanism typically have photothermal as well as photomechanical components to them, while many photochemical interactions also exhibit photothermal components. Finally, there is the possibility that multiple pulses of a particular interaction type collectively contribute to another interaction type. For example, even pulses of a duration of 100 ps, each of which have negligible thermal interactions, may add up to a measurable temperature increase if applied at repetition rates of 20 Hz or higher, depending on the laser. We will now consider each of the main interaction mechanisms in some more detail.
References:
1. Johnson, Arthur. 1999. Biological Process Engineering. New York, NY: John Wiley & Sons, Inc.
2. Incropera & DeWitt. 1990. Introduction to Conduction. New York, NY: John Wiley & Sons.
3. Niemz, M. 1996. Laser-Tissue Interaction. Berlin: Springer-Verlag
Photochemical Interactions
In the context of this module we will not go into great detail of the photochemical interactions. An excellent overview of mechanisms and some applications can be found in Niemz' book on Laser-Tissue Interaction, Chapter 3.1.
In brief, the group of photochemical interactions is based on the fact that light can induce chemical effects and reactions within macromolecules or tissues. The most obvious example of this is created by nature itself: photosynthesis. In the field of medical laser applications, photochemical interaction mechanisms play a role during photodynamic therapy (PDT). Frequently, biostimulation is also attributed to photochemical interactions, although this is not scientifically ascertained.
Photochemical interactions take place at very low irradiances (typically 1 W/cm2) and long exposure times ranging from seconds to tens of minutes. Careful selection of laser parameters (most notably wavelength) yields a radiation distribution inside the tissue that is primarily determined by scattering. In most cases, wavelength in the red and near infrared part of the spectrum (600-900 nm) are used because of their low absorption and resulting deep penetration in tissue.
During PDT chromophores (called photosensitizers) are injected in the body. Monochromatic light may then trigger selective photochemical reactions. The end result of a significant amount of complicated reactive species chemistry is that the activated photosensitizer, acting as a catalyst, causes highly cytotoxic compounds to be released causing an irreversible oxidation of essential cell structures.
Another type of photochemical laser-tissue interaction is molecular bond breaking. The absorbed photon excites the molecule to a higher electronic, vibrational, or rotational state. If the photon energy is large enough to excite the molecule to an electronic state which is not bound, the molecule dissociates. Then, it may form radicals or it may break. Clearly the prerequisite for such a mechanisms is ample supply of high energy (short wavelength, UV) photons, since only those photons posses sufficient energy to overcome the molecular bond strength of organic molecules.
Summary Photochemical Interactions:
- Main idea: using photosensitizer acting as catalyst (PDT); optical activation/modulation of certain biochemical pathways (Biostimulation)
- Observations: no macroscopic observations
- Typical lasers: red dye lasers, diode lasers
- Typical pulse durations: 1 second to CW
- Typical irradiances: 0.01 - 50 W/cm2
- Special applications: PDT, biostimulation, gene therapy (?)
Photothermal Interactions
Photothermal interactions include a large group of interaction types resulting from the transformation of absorbed light energy to heat, leading to a local temperature increase. Thermal effects can be induced by either continuous wave (CW) or pulsed laser radiation. While photochemical processes are often governed by a specific reaction pathway, photothermal effects generally tend to be non-specific and are mediated primarily by absorption of optical energy and secondly governed by fundamental principles of heat transport. Depending on the duration and peak value of the temperature achieved, different effects like coagulation, vaporization, melting or carbonization may be distinguished. Heat flows in biological tissue whenever a temperature difference exists.
The transfer of thermal energy is governed by the laws of thermodynamics.
Law #1: Energy is conserved
Law #2: Heat flows from areas of high temperature to areas of low temperature
Heat transfer is similar to fluid flow and mass transfer because it obeys the same general forms of equations. Heat transport is different from fluid flow and mass transfer in several respects: (1) no physical substance must move for heat to be transferred. (2) no fluid flow or mass transfer is analogous to radiation.
Our next task is to come up with an equation for heat transfer - i.e. we want to know the temperature distribution as function of time. How are we going to handle this??
First, let's consider what mechanisms of heat transfer there are to consider:
1) conduction
2) convection
3) radiation
4) metabolic heat
5) perfusion
We will look at these one at the time. First consider heat conduction as it will be the most significant contributor to heat transfer in laser irradiated tissue.
1) Conduction
Conduction refers to the transfer of energy in a stationary solid or fluid medium due to a temperature gradient. Direct physical contact between interacting systems is required for conduction to happen. The transfer of thermal energy occurs at the molecular and atomic levels without net mass motion of the material.
Conduction of heat is described by the heat conduction equation:
![]() (1)
(1)
To determine the temperature distribution in a medium, it is necessary to solve the appropriate form of the heat equation. Typically, the heat conduction equation written in a discrete form can be solved numerically using finite difference or finite element approaches. However, regardless on how the equation is solved, a solution depends on the physical conditions existing at the boundaries of the medium (so-called boundary conditions).
Where does this equation come from?? In the following few pages we will derive it.
Consider a control volume of tissue in Cartesian coordinates.

What sources should we consider when writing and energy balance for this control volume?
1) heat generation: laser, ultrasound, radiofrequency, metabolic, etc.
2) heat flowing into the volume
3) heat flowing out of the volume
4) heat stored in the volume
Hence we can write an energy balance for this control volume:
![]() [W or J/s] (2)
[W or J/s] (2)
With this as our starting point we first have to review some concepts of heat tranfer. Recall that heat flows from areas with a high temperature to areas with a low temperature.

As illustrated in the rod above (representing a one-dimensional conduction situation), this heat transfer rate is proportional to the area, A, the temperature gradient,![]() , and inversely proportional to the length of the rod, dx. Futhermore, there is a material constant, k [W/m K], the thermal conductivity of the material
, and inversely proportional to the length of the rod, dx. Futhermore, there is a material constant, k [W/m K], the thermal conductivity of the material
Thus, we can write the rate of heat transfer as:
![]() (3)
(3)
![]() this can be written as a differential equation, known as Fourier's law:
this can be written as a differential equation, known as Fourier's law:
 (4)
(4)
where qx is the rate of heat transfer and ![]() is the heat flux (the rate of heat transfer normalized to the area through which heat flows). The heat flux is directional (i.e. it is a vector) perpendicular to A, where A is the area of the surface with constant temperature. Furthermore the direction of the heat flow is positive when the temperature gradient is negative.
is the heat flux (the rate of heat transfer normalized to the area through which heat flows). The heat flux is directional (i.e. it is a vector) perpendicular to A, where A is the area of the surface with constant temperature. Furthermore the direction of the heat flow is positive when the temperature gradient is negative.

In general, remember that the heat flux is a vector, while T(x,y,z) is a scalar temperature field, and ![]() represents the 3-D del operator (3-D differential equation). In the above equation we only dealt with heat transfer in one dimension. However we now need to consider heat transfer in all three (x, y, and z) dimensions. Then Fourier's law can be written as:
represents the 3-D del operator (3-D differential equation). In the above equation we only dealt with heat transfer in one dimension. However we now need to consider heat transfer in all three (x, y, and z) dimensions. Then Fourier's law can be written as:
 (5)
(5)
For each of the three (x, y, and z) dimensions, the heat transfer can be written as a partial differential equation:
 (6)
(6)
Okay, we will come back to this later. Now let's go back to our little control volume. The volume, dV, itself is dx dy dz. Note that in the figure heat transfer in the y direction is not depicted for clarity of the figure.

Consider a homogenous medium with some temperature distribution, T(x,y,z) in which our control volume is situated. Conduction rates at opposite surfaces can be written using Taylor series expansion and neglecting higher order terms:
 (7)
(7)
What does this mean physically in terms that I can understand?? "The x component of the heat transfer rate at x + dx is equal to the value of this component at x plus the amount by which it changes with respect to x, times x"
Now we have done all the prework need to use our initial energy balance for the control volume. Recall that we already determined (equation (2)) that:

Substituting each of the four terms in the original energy balance equation:
![]() (8)
(8)
Substituting the Taylor series expansion for conduction rates at opposite surfaces which we derived earlier (equation (7)), results in:
![]() (9)
(9)
Recall Fourier's law (equation (4)) for each of the three dimension can be written as:
 (10)
(10)
where the heat flux is multiplied by the appropriate control surface area to get the heat transfer rate.
If we substitute this in the energy balance equation, we obtain:
![]() (11)
(11)
Since every term has the control volume, dx dy dz, in it, we can leave it out, resulting in:
 (12)
(12)
which can be rewritten as a second order differential equation (if we also divide all terms by k):
 (13)
(13)
Note that ![]() which is the thermal diffusivity of the material [m2/s].
which is the thermal diffusivity of the material [m2/s].
Another way to write this equation is:
![]() (14)
(14)
where: S = µa f is the rate of heat generation [W/cm3]
k = the thermal conductivity [W/m K]
![]() = the temperature gradient (in 3-D)
= the temperature gradient (in 3-D)
r = density [kg/m3]
c = specific heat or heat capacity [J/kg K]
![]() = the change of temperature with respect to time
= the change of temperature with respect to time
This equation is known as the heat conduction equation. Note that if the time of heating is so short that heat conduction can be neglected, the ![]() term goes to zero and which
term goes to zero and which ![]() is what we have seen before as the equation we can use to calculate the temperature rise of tissue caused by laser radiation once we know the irradiance anywhere in the tissue.
is what we have seen before as the equation we can use to calculate the temperature rise of tissue caused by laser radiation once we know the irradiance anywhere in the tissue.
Well that is it for heat conduction for now (what a pain to type in equation editor….).
Next, let's consider the other mechanisms of heat transfer:
2) Convection
Convection is the transfer of thermal energy through a fluid due to bulk motion of the fluid. Note that a fluid can be a gas or liquid). It typically involves four intermediate processes:
a. Conduction of heat from the solid surface to the immediately adjacent fluid particles.
b. Absorption and storage of the conducted energy by these fluid particles with the elevation of their internal energy
c. Migration of these higher-energy fluid particles to regions of lower temperature, with mixing and transfer of part of the stored energy
d. Transport of the energy by bulk movement of the fluid
The convective cooling effect via blood circulation is an important factor in determining the tissue temperature distribution. In many cases of laser heating, the contribution of blood flow through the capillary bed can be neglected without introducing appreciable error. On the other hand, when a large blood vessel passes through the heated region or following the period of irradiation, convection can be a much more effective heat transfer mechanism than conduction. We will consider how to include perfusion in our temperature distribution calculations shortly.
Convection is described by Newton's law of cooling which states that:
![]() (15)
(15)
where: h is the convection coefficient [W/m2 K]
Tsurface is the surface temperature of the heated material (tissue)
is the temperature of the convective environment
For example:
hdry skin = 2 10-4 [cal/cm2 s °C]
hwet skin = 7 10-4 [cal/cm2 s °C]
3) Radiation
Radiation is defined as thermal energy transfer via electromagnetic wave action between noncontacting systems at different temperatures. Unlike either conduction or convection, no medium is required for energy transport. Thermal radiation occupies the portion of the electromagnetic wave spectrum corresponding to the wavelength band of 0.1 to 10 micrometers.
Radiation is described by the Stefan-Boltzman law which states:
![]() (16)
(16)
where: e is the emissivity of the material, ![]() (typically: 0.9 - 0.95 for tissue)
(typically: 0.9 - 0.95 for tissue)
ø is the Stefan-Boltzman constant, 5.67 10-8 [W/m2 K4]
Tsurface is the surface temperature of the heated material (tissue)
![]() is the temperature of the environment
is the temperature of the environment
The term emissivity is sometimes also referred to as 'black body radiation'. An ideal 'black body' has an emissivity of 1 and has the characteristics that it
1) absorbs all radiation regardless of wavelength or direction, and
2) emits radiation diffusely.
e = 1 ------- blackbody
e = 0 ------- complete reflection
Another way of saying this is that a so-called blackbody is a perfect emitter radiating the maximum amount of radiant energy at any wavelength for a given emitter temperature. It is also a perfect absorber reflecting no thermal energy. That is why a black dog will feel hotter than a white dog if both are the same temperature.
These (conduction, convection and radiation) are the three common mechanisms of heat transfer. In medical applications and in particular laser heating of tissue, we may also have to consider metabolic heat, and perfusion (i.e. cooling due to the fact that blood carries heat away from the irradiated volume).
Note: Blackbody radiation is used in infrared imaging to detect heat emitting objects (night vision goggles, heat seeking missiles, thermal cameras, etc.). Generally all these infrared imagers are instruments that create pictures of heat rather than light. They measure radiated blackbody radiation and convert the data to corresponding maps of temperatures. The thermal camera is calibrated using IR radiation power vs. temperature curves. The information is then digitized and displayed on a monitor.


4) Metabolic heat
Metabolic heat is typically not a significant factor in thermal laser interactions. However, for long exposures in particular this term may add to the energy in the irradiated volume. However in most cases, this is taken care of by assuming that the starting temperature of the irradiated tissue is approximately 37 °C.
5) Perfusion
A model of convective heat transfer in biological tissue has to consider the blood flow distribution of the perfused tissue. The perfusion distribution markedly influences the local temperature. For example, in the extreme case of no blood flow in exercising muscle, tissue temperature may rise as high as 45°C. Introduction of flowing blood at normal rates, such as 1 ml/g min, causes temperature to promptly drop to physiologically compatible values. When blood perfusion rate is allowed to increase to fivefold normal values, the temperature of the tissue becomes uniform and equal to arterial temperature.
For longer exposures (several seconds and longer), the fact that blood flows in and out of the irradiated volume may have a significant effect on the heat balance of the irradiated volume. Body temperature arterial blood (37 oC) flows into the irradiated volume which will have a higher temperature. Conduction and convection and even direct radiation of the blood by the laser will cause the temperature of the departing blood to be higher. In the heat balance this is a loss term:
![]() (17)
(17)
The perfusion term is difficult to know exactly since it depends on the amount of blood perfusion of the tissue. In addition heating is known to cause vasodilation while excessive heating can cause coagulation (and thus reduced blood flow) and hence the perfusion is not constant. Nevertheless, a first order approximation can be formulated based on the application of Fick's principle, "the amount of substance taken up by an organ per unit time is equal to the arterial level of the substance minus the venous level time the rate of blood flow":
![]() (18)
(18)
where:
wb is the volume fraction of the tissue consisting of blood that is replaced per

second, i.e. the perfusion rate
rb is the density of blood [kg/m3]
cb is the specific heat of blood [J/kg K]
Tvenous is the temperature of the venous blood (leaving the irradiated tissue)
Tarterial is the temperature of the arterial blood (entering the irradiated tissue)
In this expression we assume a uniform cooling / heating of dV by the capillary bed. In summary, the perfusion term should be considered when long exposure times are involved (several seconds or longer), depending on the perfusion rate of the tissue under consideration.
6) Heat of vaporization
Finally, when we heat tissue there is one more term which we have ignored thus far, to which energy may be lost in large amounts, and that is phase changes (vaporization). This term can be quite significant. For example, looking at the energy required to boil water (and tissue is 70 - 80% water), the energy required to heat water from room temperature to 100 °C is roughly one tenth of the energy required to cause water to become water vapor (known as the latent heat of vaporization):
![]() (19)
(19)
where![]()
and![]()
Summary Photothermal Interactions:
- Main idea: achieving a certain temperature which leads to the desired thermal effect
- Observations: either coagulation, vaporization, melting, or carbonization
- Typical lasers: CO2, Nd:YAG, Ho:YAG, Er:YAG, Argon ion, diode lasers
- Typical pulse durations: 1 ms - minutes
- Typical irradiances: 10 - 106 W/cm2
- Special applications: coagulation, vaporization, melting, thermal decomposition, treatment of retinal detachment, Port-wine stain, retinal photocoagulation, interstitial hyperthermia
Photomechanical Interaction
Photomechanical interaction includes mechanisms associated with photoablation, explosive vaporization, plasma induced ablation, and effects of laser induced acoustic (shock waves). Photomechanical mechanisms can be caused by the thermoelastic expansion of tissue due to extremely rapid heating of the target tissue by the pulsed laser light and recoil caused by ejection of ablated material. These photomechanical interactions cause stress waves, which propagate with the speed of sound or faster. If the laser pulse length is shorter than the time it takes the stress wave to propagate out of the irradiated tissue volume, large peak stresses can be reached. These stresses may contribute to the ablation mechanism and may inflict damage to adjacent tissue. Since virtually all photomechanical interactions lead to ablation, we will discuss of them in more detail in the next module on ablation.
