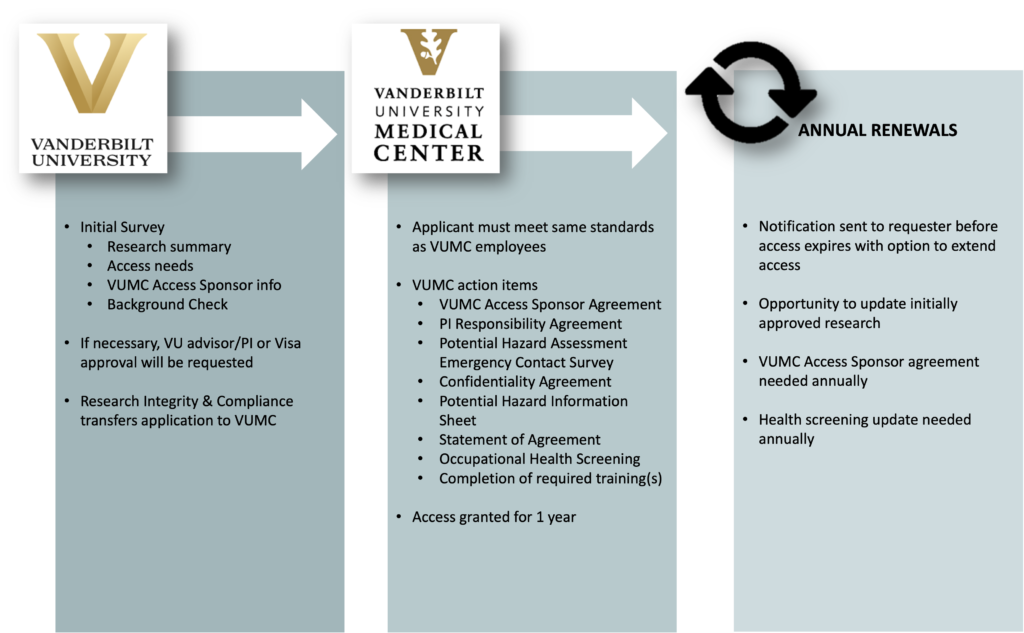
Researchers across Vanderbilt University (VU) may need access to Vanderbilt University Medical Center (VUMC) clinical systems, facilities or patients (VUMC Resources) to complete their VUMC IRB-approved or VUMC IRB-ceded research projects. To support this important work, VU has an agreement with VUMC to allow approved VU-employed researchers, VU post-doctoral appointees, and VU advanced academic degree students access to VUMC Clinical Resources.
To request access to VUMC Clinical Resources, please complete the initial survey found here. Once submitted, a member of our team will review your responses and contact you if additional information is required. For questions or assistance, please email ric@vanderbilt.edu.