3 – Metabolomics Sample Storage and Handling
In an effort to generate the highest quality data and observe the most complete metabolite profile, the tips for metabolomics sample storage and handling conditions outlined below are aimed at preserving the metabolites present at the time of sampling. To this end,it is necessary to prevent sample degradation by minimizing pH, temperature, or oxidation fluctuations; to avoid sample contamination, and to minimize residual enzymatic activity (metabolism quenching).
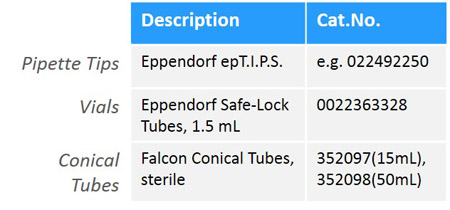
Materials for Sample Collection
We suggest storing samples in small aliquots to avoid recurring freeze-thaw cycles, which can negatively affect sample quality. In our experience, we have observed the best results/highest data quality by using the following lab materials for sample handling and storage:
If you are using different materials for sample collection from the ones above, evaluation of the collection containers is recommended. In general, contact of samples with poly(ethylene glycol) materials should be minimized, if possible, as they can leach contaminants into your samples.
Sample Type-Specific Handling Recommendations
Cells (for intracellular metabolites)
Methods for effective cell disruption or lysis for metabolite identification differ depending on the specific cell type. For best results, we suggest rinsing cells quickly with ice-cold 50mM ammonium formate (pH6.8-7.0). After removing media from the cells, please rapidly freeze in liquid nitrogen followed by storage at -80ºC. Please refrain from using detergents or buffers (e.g. PBS) on your samples as the salts (NaCl, etc.) interfere with future analyses. Upon request, the CIT can provide 50 mM ammonium formate at pH 6.8-7.0.
Cell Effluent (for extracellular metabolites)
Please avoid cell lysis and consequential intracellular metabolite leakage by careful handling of the cell culture. Cell or tissue media should be centrifuged at low speed (to remove cellular debris or cells) and supernatant removed prior to freezing. For best results, we suggest rapidly freezing samples in liquid nitrogen followed by storage at -80ºC.
Cerebrospinal Fluid (CSF)
Signal intensity for CSF is lower than other biofuids, which requires especially careful handling and storage. We recommend centrifugation immediately after sampling and sample storage at -80ºC. Please also be careful to avoid blood contamination of the CSF sample.
Plasma
The addition of heparin, citrate, or EDTA to prevent fibrinogen clotting will require extremely careful reporting of metabolites in the anti-coagulant retention time window. Please note that different coagulants will have different retention time peaks. To minimize hemolysis and therefore prevent release of intracellular metabolites, please centrifuge at low speed to isolate the supernatant. Samples should be stored at -80ºC and freeze-thaw cycles avoided.
Serum
Serum offers a greater sample volume than plasma for volume of blood drawn, which results in improved accuracy for low abundance metabolites. Some metabolic processes take less than a second. To minimize residual metabolic activity, please consider to conduct clotting procedure on ice instead of room temperature. To minimize hemolysis and therefore prevent release of intracellular metabolites, please centrifuge at low speed to isolate the supernatant. Samples should be stored at -80ºC and freeze-thaw cycles avoided.
Solid Tissues
It is recommended to rinse solid tissues (w/ ice-cold 50mM ammonium formate) to remove blood prior to storage in liquid nitrogen for metabolic quenching. Please refrain from using detergents or buffers (e.g. PBS) on your samples as this interferes with our analysis. Upon request, the CIT can provide 50mM ammonium formate at pH 6.8-7.0.
Urine
While sodium azide may be used as bacteriostatic agent during sample storage, 0.2 μm filtration may be preferred unless sample volume is very limited. Mild pre-centrifugation at 1000-3000 rcf for 5 minutes is recommended to remove cellular matter without inducing cell breakage and resultant release of intracellular metabolites. Samples should be stored at -80ºC and freeze-thaw cycles avoided.
Additional sample-type recommendations are under development. Please contact us if you have any questions.



 Follow Us
Follow Us