Hillyer Lab News

Article describing the myotropic effects of mosquito FMRFamide containing peptides is published in General and Comparative Endocrinology
Friday, April 18, 2014

The tetrapeptide, FMRFamide, was first discovered because of its cardioacceleratory activity in a crab. Since then, FMRFamide containing peptides have been identified in a broad range of invertebrate and vertebrate animals and shown to modulate numerous physiological processes. As pertains to cardiac physiology in insects, different FMRFamide containing peptides are known to (1) be cardioacceleratory, (2) be cardiodeceleratory, (3) have complex effects that are dependent on the presence of other molecules, and (4) have no effect on heart physiology.
In this newly published study, we describe the structure of the FMRFamide gene in the mosquito Anopheles gambiae, assess the developmental expression of FMRFamide and the putative FMRFamide receptor (FMRFamideR), and describe the the effects of the peptides FMRFamide and SALDKNFMRFamide on mosquito heart physiology. Specifically, we show that:
-
1. The FMRFamide gene is alternatively spliced and encodes seven different putative RFamide containing peptides (one is encoded twice). Of these, only one contains an FMRFamide sequence (SALDKNFMRFamide), and this peptide is the only one that is conserved across the mosquito lineage (Anopheles gambiae, Aedes aegypti and Culex quinquefasciatus).
-
2.The developmental expression of FMRFamide and FMRFamideR displays a bimodal distribution, with expression peaks occurring in second instar larvae and around eclosion (adult emergence).
-
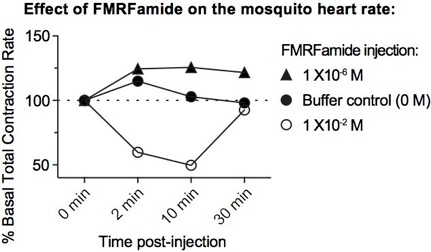
3.Exposure to low doses of the peptides FMRFamide or SALDKNFMRFamide leads to an increase in heart contraction rates (see figure).
-
4.Exposure to high doses of the peptides FMRFamide or SALDKNFMRFamide leads to a decrease in heart contraction rates and a biasing of the proportional directionality of heart contractions (see figure).
-
5.In conclusion, these data show that FMRFamide containing peptides are complex modulators of mosquito heart physiology.
ARTICLE CITATION:
Hillyer, J.F., T.Y. Estévez-Lao, and L.E. de la Parte. 2014. Myotropic effects of FMRFamide containing peptides on the heart of the mosquito Anopheles gambiae. General and Comparative Endocrinology. 202:15-25.
(Pubmed) (See it in GCE) (Email me for a pdf copy)
GRAPHICAL ABSTRACT:
ARTICLE ABSTRACT:
FMRFamide-like peptides (FLPs) are produced by invertebrate and vertebrate animals, and regulate diverse physiological processes. In insects, several FLPs modulate heart physiology, with some increasing and others decreasing dorsal vessel contraction dynamics. Here, we describe the FMRFamide gene structure in the mosquito, Anopheles gambiae, quantify the developmental and spatial expression of FMRFamide and its putative receptor (FMRFamideR), and show that the peptides FMRFamide and SALDKNFMRFamide have complex myotropic properties. RACE sequencing showed that the FMRFamide gene encodes eight putative FLPs and is alternatively spliced. Of the eight FLPs, only one is shared by A. gambiae, Aedes aegypti and Culex quinquefasciatus: SALDKNFMRFamide. Quantitative PCR showed that peak expression of FMRFamide and FMRFamideR occurs in second instar larvae and around eclosion. In adults, FMRFamide is primarily transcribed in the head and thorax, and FMRFamideR is primarily transcribed in the thorax. Intravital video imaging of mosquitoes injected FMRFamide and SALDKNFMRFamide revealed that at low doses these peptides increase heart contraction rates. At high doses, however, these peptides decrease heart contraction rates and alter the proportional directionality of heart contractions. Taken altogether, these data describe the FMRFamide gene in A. gambiae, and show that FLPs are complex modulators of mosquito circulatory physiology.
(This post was modified on 5/14/2014 to include the full citation of the article)
General and Comparative Endocrinology publishes articles concerned with the many complexities of vertebrate and invertebrate endocrine systems at the sub-molecular, molecular, cellular and organismal levels of analysis.


